NORMAL UMBILICAL CORD MATRIX
|
- The normal matrix consists of Wharton's jelly, which binds and encases the umbilical vessels, protecting them from twisting and compression during pregnancy and delivery. It is composed of collagen fibers forming a network of interconnected cavities, cavernous and perivascular spaces in which the ground substance of the jelly is stored (1-15).
- The Whartons jelly mainly comprises:
- A ground substance of hyaluronic acid and proteoglycans in an aqueous solution of salts, metabolites and plasma proteins distributed in a fine network of collagen microfibrils (11).
- Cellular population consists predominantly of fibroblasts involved in synthesizing collagen and glycosaminoglycans (12).
- Several types of collagen (types I, III, IV, V
and VI) are homogeneously distributed in the media of the umbilical vessels
or in the Whartons jelly (13,14).
- Approximately 70% of the soluble part of the
Whartons jelly is composed of collagen type IV and hyaluronic acid (12)
(which is capable of entrapping large amounts of fluid (15).
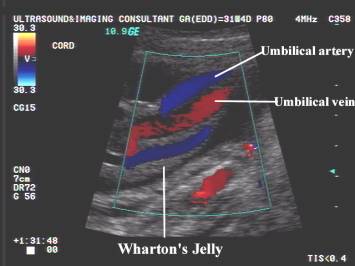
- Wharton's jelly develops from the extraembryonic mesoderm and provides mechanical support and structural protection for the umbilical vessels. It also has angiogenic and metabolic roles for the umbilical circulation (2).
- Both pediatricians and pathologists have known for many years that the amount of Wharton's jelly is a good predictor of perinatal complications.
- The most common macroscopic finding of the modifications of WJ composition is variation in umbilical cord size.
- A reduced amount of Wharton's jelly in an otherwise normal cord has been associated with an increased perinatal mortality (fetal distress, growth restriction and oligohydramnios).
- Changes or alterations of any of the WJ components have been described or postulated in some pathological conditions such as hypertensive disorders (3), fetal distress (4), gestational diabetes (5,6) and fetal growth restriction (7).
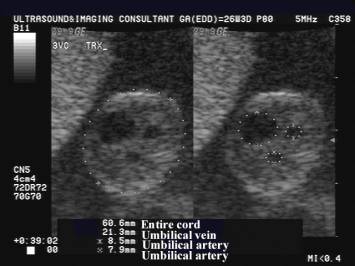
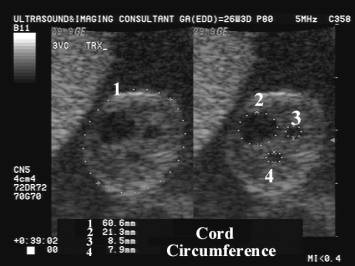
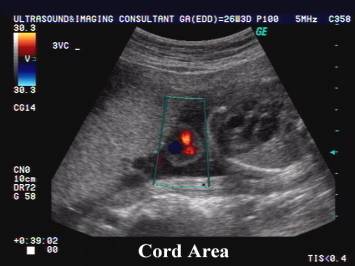
- The umbilical cord cross-sectional area and diameter measured sonographically has been correlated with fetal anthropometric parameters (8). The detection of a lean cord in the second half of gestation has been found associated with the delivery of a small-for-gestational age infant and with an increased risk of fetal distress during labor (9). A large umbilical cord diameter without alteration of the vessels’ diameter has been reported in pregnancies complicated by gestational diabetes (10).
REFERENCES |
- Klein J & Meyer FA. Tissue structure and macromolecular diffusion in umbilical cord. Immobilization of endogenous hyaluronic acid. Biochim Biophys Acta 1983; 22: 400–11
- Benirschke K, Kaufmann P, (eds). Umbilical cord and major fetal vessels. In: Pathology of the human placenta, 2nd edn. New York: Springer - Verlag 1990:180-243.
- Bankowski E, Sobolewski K, Romanowicz Let.al. Collagen and glycosaminoglycans of Wharton’s jelly and their alterations in EPH-gestosis. Eur J Obstet Gynecol Reprod Biol 1996; 66: 109–117
- Goodlin RC. Fetal dysmaturity, ‘lean cord’, and fetal distress. Am J Obstet Gynecol 1987; 156: 716
- Singh SD. Gestational diabetes and its effect on the umbilical cord. Early Hum Dev 1986; 14: 89–98
- Ali FMA, Fateen B, Ezzet A.et.al. Lack of proteoglycans in Wharton’s jelly of the human umbilical cord as a cause of unexplained fetal loss in diabetic infants. Obstet Gynecol 2000; 95: 61S
- Bruch JF, Sibony O, Benali K et.al. Computerized microscope morphometry of umbilical vessels from pregnancies with intrauterine growth retardation and abnormal umbilical artery Doppler. Hum Pathol 1997; 28: 1139–1145
- Raio L, Ghezzi F, Di Naro E et.al. Sonographic measurements of the umbilical cord and fetal anthropometric parameters. Eur J Obstet Gynecol Reprod Biol 1999; 83: 131–135
- Raio L, Ghezzi F, Di Naro Eet.al. Prenatal diagnosis of a ‘lean’ umbilical cord: a simple marker for fetuses at risk of being small for gestational age at birth. Ultrasound Obstet Gynecol 1999; 13: 76–80
- Weissman A & Jakobi P. Sonographic measurements of the umbilical cord in pregnancies complicated by gestational diabetes. J Ultrasound Med 1997; 16: 691–694
- Takechi K, Kuwabara Y, Mizuno M. Ultrastructural and immunohistochemical studies of Wharton’s jelly umbilical cord cells. Placenta 1993; 14: 235-245
- Vizza E, Correr S, Goranova V, Heyn R, Angelucci PA, Forleo R, Motta PM. The collagen skeleton of the human umbilical cord at term. A scanning electron microscopy study after 2N-NaOH maceration. Reprod Fertil Dev 1996; 8: 885-894
- Nanaev AK, Kohnen G, Milovanov AP, Domogatsky SP, Kaufmann P. Stromal differentiation and architecture of the human umbilical cord. Placenta 1997; 18: 53-64
- Von Kaisenberg CS, Krenn V, Ludwig M, Nicolaides KH, Brand-Saberi B. Morphological classification of nuchal skin in human fetuses with trisomy 21, 18 and 13 at 12-18 weeks and in a trisomy 16 mouse. Anat Embryol (Berl) 1998; 197: 105-124
- Klein J, Meyer F. Tvolume
structure and macromolecular diffusion in umbilical cord immobilization of
endogenous hyaluronic acid. Biochim
Biophys Acta 1983; 22:
400-411