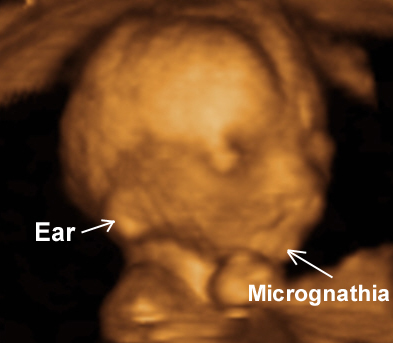
|
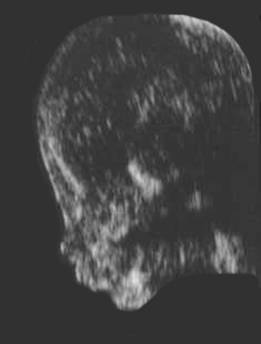
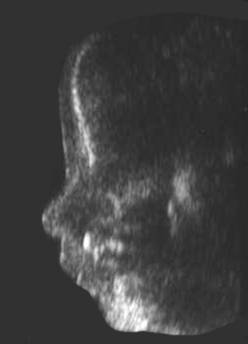
NORMAL / ABSENT / SHORT NASAL BONE |
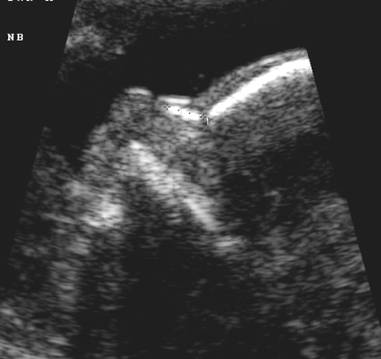
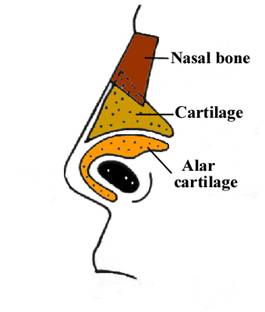
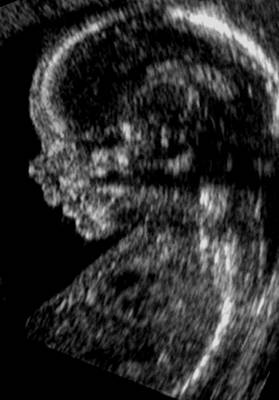
The right and left nasal bones are joined by a thick synostosis that extends from the nasal root just below the glabella to the tip of the ossified part of the nose.
The synostosis is hyperechogenic when compared to the thin distal cartilaginous portion extremity of the nose that is nearly echolucent.
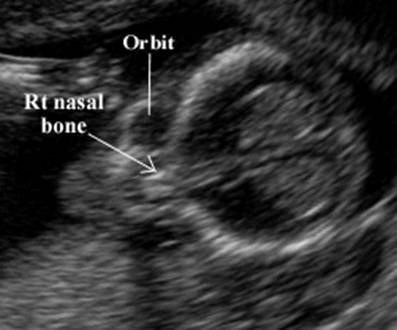
The nasal bones are trapezoidal in shape with the inner edge being shorter than the outer edge.
The nasofrontal fontanelle, or fonticulus frontalis,
temporarily separates the embryonic nasal and frontal
bones. Simultaneously,
the transient prenasal
space separates the nasal
bones and the
cartilaginous nasal
capsule. A diverticulum of dura mater extends from the anterior
cranial fossa through the foramen cecum into the transient prenasal space. It briefly
contacts the skin at the tip of the nose before retracting back into
the cranium. The tract of the dural diverticulum quickly involutes. The
nasal and
frontal bones fuse, obliterating
the fonticulus frontalis and forming the nasofrontal suture. The prenasal space becomes
smaller with growth of the adjacent bone structures, eventually
being reduced to a small canal anterior to the crista galli known as
the foramen cecum. Finally, the foramen cecum is filled with fibrous
tissue and fuses with the prenasal
space.
The nasal bridge, or glabella, is superficial to the nasal and frontal bones. A midline separation between the paired nasal bones is normally seen and should not be mistaken for a dermal sinus tract. The cribriform plate separates the nasal cavity from the anterior cranial vault.
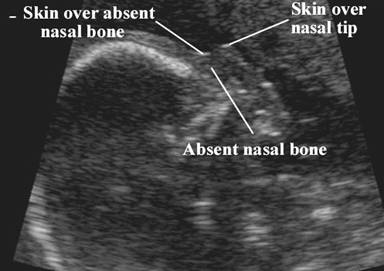
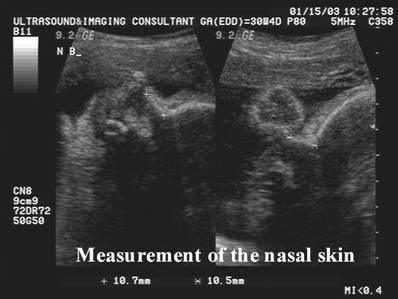
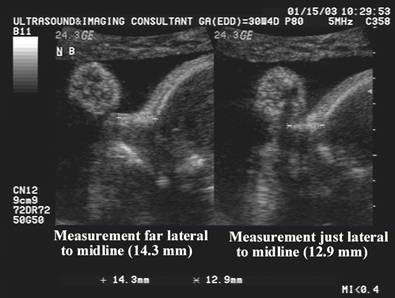
· The nose bone should be visualized on ultrasound along the mid-sagittal plane of the, the face, as the inner edge of the nasal bone is shorter than the outer edge due to the trapezoidal shape of the nasal bone. (a measurement more laterally would overestimate the size and overlook hypoplasia) (1).
· The fetal neck should be in slight flexion.
· The fetal spine should be facing downward.
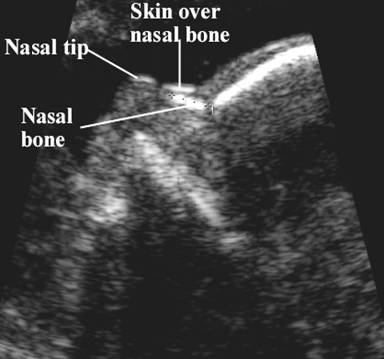
· Three echogenic lines at the fetal nose bone profile should be evident:
1. a superficial line overlying the nasal tip.
2. a second superficial line overlying the nasal bone.
3. a deeper echogenic line representing the nasal bone, which is also more echolucent at the distal end.
· The ultrasound beam should not be parallel to the plane of the nose bone, because it may erroneously suggest an absent nose bone.
· An increase in the length of the nasal bone throughout pregnancy parallels growth of other fetal parameters especially femur length.
·
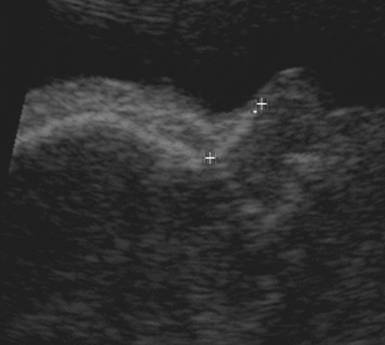
Measure the hyperechoic synostosis that joins
the left and right nasal bones.
|
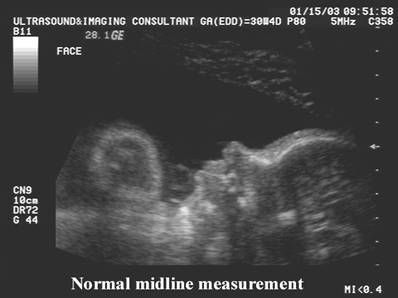
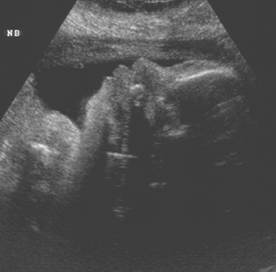
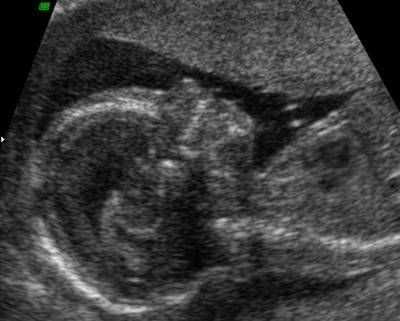
Normal
Nasal Bone |
|
|
|
|
|
|
|
|
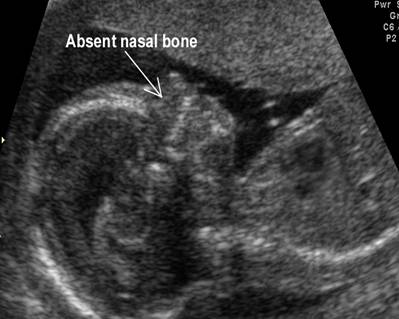
Absent
nasal bone |
|
|
|
|
|
NASAL BONE – FIRST TRIMESTER |
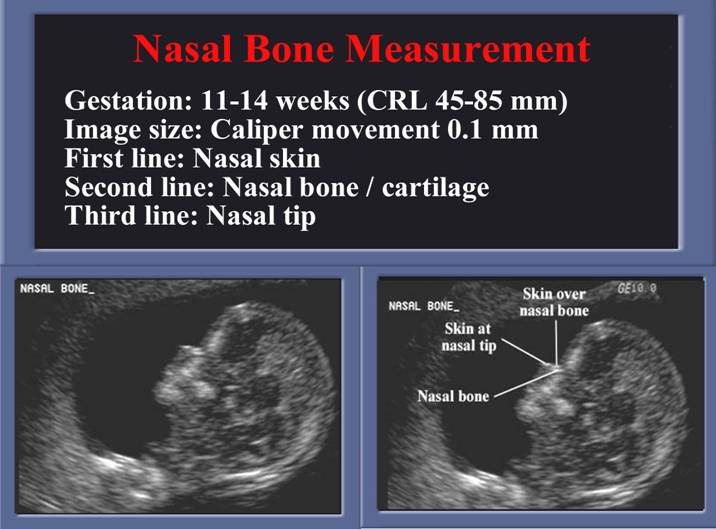
First Trimester: Measurement not necessary only it’s presence.
Fetal
nasal bone length in chromosomally normal and abnormal fetuses at 11-14 weeks
of gestation.
Cicero S, Bindra R, Rembouskos G, Tripsanas C, Nicolaides KH.
The fetal profile was examined and the nasal bone length was measured in 1092
fetuses immediately before chorionic villous sampling for karyotyping at 11-14
weeks of gestation.
· The median gestation was 12 (11-14) weeks.
· The fetal profile was successfully examined in all cases.
· The fetal karyotype was normal in 955 pregnancies and abnormal in 137, including 79 cases of trisomy 21.
· In the chromosomally normal group, the fetal nasal bone length increased significantly with crown-rump length (CRL) from a mean of 1.3 mm at a CRL of 45 mm to 2.1 mm at a CRL of 84 mm.
· In 54 of the 79 (68.4%) cases of trisomy 21, the nasal bone was absent.
· In the 25 cases with present nasal bone, the nasal bone length for the CRL was not significantly different from normal. Similarly, there were no significant differences from normal in the nasal bone length of fetuses with other chromosomal defects.
· CONCLUSIONS: At 11-14 weeks of gestation, the nasal bone length of chromosomally abnormal fetuses is not significantly different from normal. The authors concluded ‘the absence of a nasal bone is a powerful marker for Down syndrome. A short nasal bone is associated with an increased likelihood for fetal Down syndrome in a high-risk population.’ They suggested that the absence of a nasal bone, even when it is the only ultrasound marker, should prompt the physician to offer an amniocentesis for fetal karyotyping.
|
NASAL BONE – SECOND TRIMESTER |
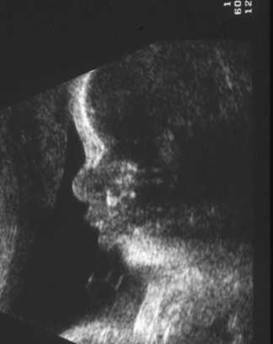
- A strict median sagittal view remains the key plane for assessing the morphology of the nasal bone.
- On a sagittal images defined structures from top to bottom include:
- Frontal bone.
- Nasal notch or bridge.
- Ossified portion of the nasal bone.
- Lips and maxilla.
- Lower lip.
- Manndible.
- Measure echogenic central synostosis.
- Medial profile essential (visualized structures include nasal synostosis, upper and lower lips, aligned maxilla and mandible).
- The nasal bone initially appears as a thin echogenic structure at a distance from the frontal bone.
- In the coronal plane the two bilateral echogenic nasal bones may be observed.
|
|
|
- The normal range is provided by the length of the synostosis joining the inner edges of the left and right nasal bone.
- A measurement too lateral would overestimate the nasal bone and may overlook nasal hypoplasia.
|
|
|
|
|
|
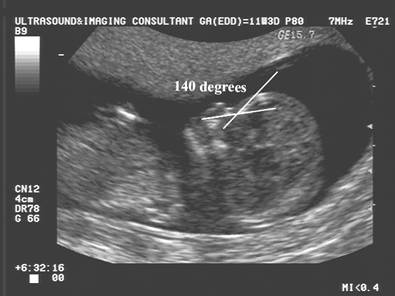
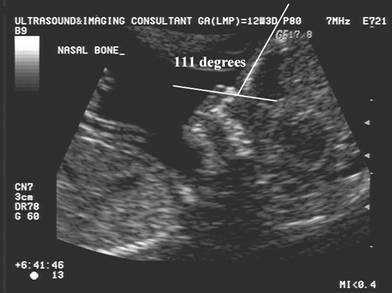
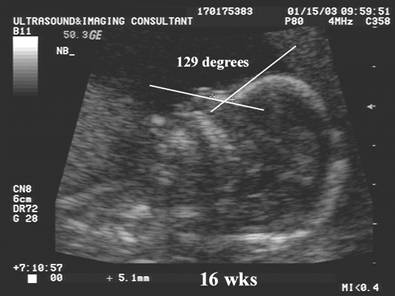
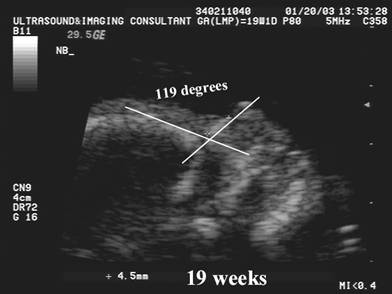
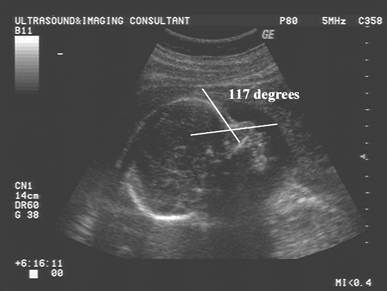
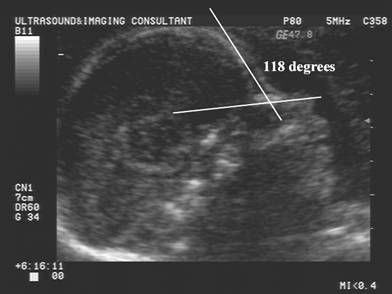
Naso-frontal
angle:
- Measurement of the angle between the frontal bone and inferior aspect of the nasal bone.
- Normal = Guis 128.60 +/- 230
- Down syndrome – Guis reported no significant difference from normal.
|
|
|
|
|
|
|
|
|
|
ABSENCE OF NASAL BONE IN FETUSES WITH TRISOMY 21 (2) |
- Absent ossification of nasal bone.
- Delayed ossification of nasal bone.
- Hypoplasia of nasal bone.
- Stempfle et.al. studied the ossification of the nasal bone in fetuses with and without DS. On postmortem radiographs, ossification of the NB is present from 15 weeks of gestation in normal fetuses. By contrast, 14/60 trisomic fetuses (23%) had no nasal none ossification at any time in the gestation. In fetuses in whom the nasal bone was ossified they tended to be shorter than did euploid fetuses.
- Keeling et al investigated axial radiographs in fetuses between 12 and 24 weeks gestation and reported agenesis of the nasal bone in 6% of fetuses with DS. Malformations of the nasal bone such as a short nose was seen in another 11 of 31 fetuses. Overall 19/31 fetuses (61%) with Ds had either absent or short nasal bones.
- Bromley et,al, studied the ultrasound characteristics of 16 fetuses with Down syndrome and compared them to 223 euploid fetuses.
o
In
fetuses with Down syndrome, 6 (37%) of 16 did not have detectable nose bones
compared with 1 (0.5%) of 223 control fetuses.
o
For
the control group the BPD/NBL ratio was constant during the 15–20 week
period of gestational age bring studied.
o
Using
a cutoff of BPD/NBL ratio of 9 or greater resulted in 100% detection of Down
syndrome but a 22% detection rate in the control group.
o
If
a cutoff were raised to 11 or greater, then 69% of fetuses with Down syndrome
would be identified compared with 5% of euploid fetuses, yielding a likelihood
ratio of 11.8.
o
Of
the twelve euploid fetuses with BPD/NBL ratio 11 or greater one showed other
features consistent with an unidentified syndrome, and four showed short
femurs.
o In total, results
demonstrated that 61% of fetuses with Down syndrome displayed either an absent
or short nasal bone. A short or absent nasal bone was the only ultrasound
marker for aneuploidy in three fetuses with Down syndrome.
·
A recent study was undertaken by
researchers at King’s
·
The crown rump length ranged from
45–84 mm, which is in keeping with that suggested for nuchal translucency
screening.
·
A fetal profile that enabled
establishment of the presence of absence of the nasal bone was obtained in all
cases. The optimal plane for nasal bone imaging was a mid-sagittal section with
the sound beam perpendicular to the nasal bone. The authors warned that care
should be taken not to confuse the echogenic skin surface of the nose with the
nasal bone.
·
This study has shown that, at 11 –
14 weeks of gestation:
o
The nasal bone is visible by
ultrasonography in 99.5% of chromosomally normal fetuses.
o
The nasal bone was absent in 73% of trisomy
21 fetuses (and in 0.5% of chromosomally normal fetuses), which could be due to
hypoplasia or deferred ossification.
o
The likelihood ratio for trisomy 21 was
146 (95% CI 50,434) for absent nasal bone and 0.27 (95% CI 0.18, 0.4) for
present nasal bone.
o
This study suggests that sensitivity
would be increased from 75% to 85% and that the false positive rate would
decrease from 5% to 1% when the fetal profile was included with maternal age
and NT measurement.
o
They interpreted that ‘in screening
for trisomy 21, examination of the fetal nasal bone could result in major
reduction in the need for invasive testing and a substantial increase in
sensitivity’.
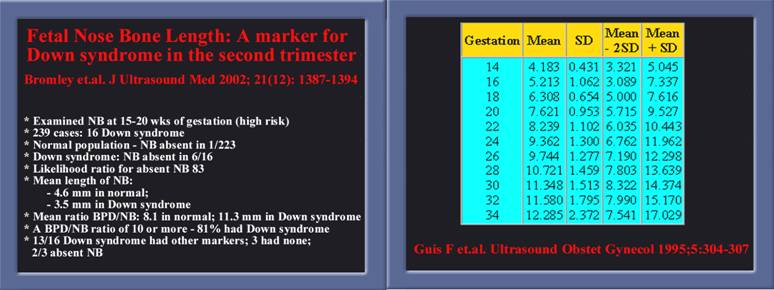
- Small or hypoplastic nose is found in many syndromes including Trisomy 21 and 18, Apert’s Syndrome, de Lange syndrome and fetal toxins.
Normal Nasal Bone
|
||
|
|
|
|
Down Syndrome
|
||
|
Mild Nasal Hypoplasia |
Marked Nasal Hypoplasia |
Absent Nasal Bone |
|
|
|
|
|
|
|
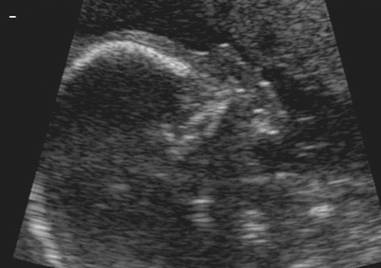
Absent nasal bone at 32 wks |
· Absent nasal bone and Fragile X Syndrome.
· Nasal bone may be absent in trisomy 18.
|
Trisomy 18
– Absent nasal bone |
|
|
|
|
|
|
|
REFERENCES |
- Guis F, Ville Y, Doumerc S et al: Ultrasound evaluation of the length of the fetal nasal bones throughout gestation. Ultrasound Obstet. Gynecol. 1995, 5: 304-307.
- Cicero S, Curcio P, Papageorghiou A et.
al. Absence of nasal bone in fetuses with trisomy
21 at 11–14 weeks of gestation: an observational study. Lancet
2001(November); 358:1665–67